  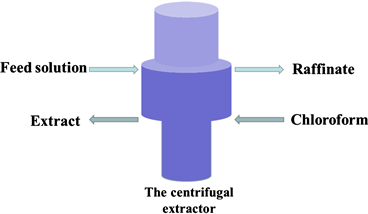
Note that D is related to the Δ G of the extraction process. Depending on the system, the distribution ratio can be a function of temperature, the concentration of chemical species in the system, and a large number of other parameters. The distribution ratio ( K d) is equal to the concentration of a solute in the organic phase divided by its concentration in the aqueous phase. In solvent extraction, a distribution ratio is often quoted as a measure of how well-extracted a species is. ( May 2014) ( Learn how and when to remove this template message) Unsourced material may be challenged and removed. Please help improve this article by adding citations to reliable sources in this section. This section needs additional citations for verification. A detergent or fine solid can be used to stabilize an emulsion, or third phase. For example, it is possible for sodium cations to be reduced at a mercury cathode to form sodium amalgam, while at an inert electrode (such as platinum) the sodium cations are not reduced. This is related to a mercury electrode where a metal can be reduced, the metal will often then dissolve in the mercury to form an amalgam that modifies its electrochemistry greatly. Liquid–liquid extraction is possible in non-aqueous systems: In a system consisting of a molten metal in contact with molten salts, metals can be extracted from one phase to the other. It is among the most common initial separation techniques, though some difficulties result in extracting out closely related functional groups. LLE is also widely used in the production of fine organic compounds, the processing of perfumes, the production of vegetable oils and biodiesel, and other industries. Stripping is the opposite of extraction: Transfer of mass from organic to aqueous phase. One obtains high-purity single metal streams on 'stripping' out the metal value from the 'loaded' organic wherein one can precipitate or deposit the metal value. įrom a hydrometallurgical perspective, solvent extraction is exclusively used in separation and purification of uranium and plutonium, zirconium and hafnium, separation of cobalt and nickel, separation and purification of rare earth elements etc., its greatest advantage being its ability to selectively separate out even very similar metals. In that case, a soluble compound is separated from an insoluble compound or a complex matrix. The term solvent extraction can also refer to the separation of a substance from a mixture by preferentially dissolving that substance in a suitable solvent. The term partitioning is commonly used to refer to the underlying chemical and physical processes involved in liquid–liquid extraction, but on another reading may be fully synonymous with it. This type of process is commonly performed after a chemical reaction as part of the work-up, often including an acidic work-up. LLE is a basic technique in chemical laboratories, where it is performed using a variety of apparatus, from separatory funnels to countercurrent distribution equipment called as mixer settlers. The feed solution that is depleted in solute(s) is called the raffinate. The solvent that is enriched in solute(s) is called extract. once the transfer is complete, the overall system of chemical components that make up the solutes and the solvents are in a more stable configuration (lower free energy). The transfer is driven by chemical potential, i.e. There is a net transfer of one or more species from one liquid into another liquid phase, generally from aqueous to organic. Liquid–liquid extraction ( LLE), also known as solvent extraction and partitioning, is a method to separate compounds or metal complexes, based on their relative solubilities in two different immiscible liquids, usually water (polar) and an organic solvent (non-polar).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
